CRISPR-Cas9: The Double-Edged Sword of Gene Editing
In the annals of scientific breakthroughs, few discoveries have generated as much simultaneous excitement and trepidation as CRISPR-Cas9. This revolutionary gene-editing tool, adapted from a naturally occurring defense mechanism in bacteria, has provided scientists with an unprecedented ability to alter the DNA of living organisms with remarkable precision and ease. Its potential applications are staggering, ranging from the eradication of genetic diseases to the enhancement of agricultural crops. However, this immense power is a double-edged sword, compelling a profound and urgent societal reckoning with the ethical, social, and philosophical ramifications of rewriting the very code of life.
The Mechanism: A Molecular Scalpel
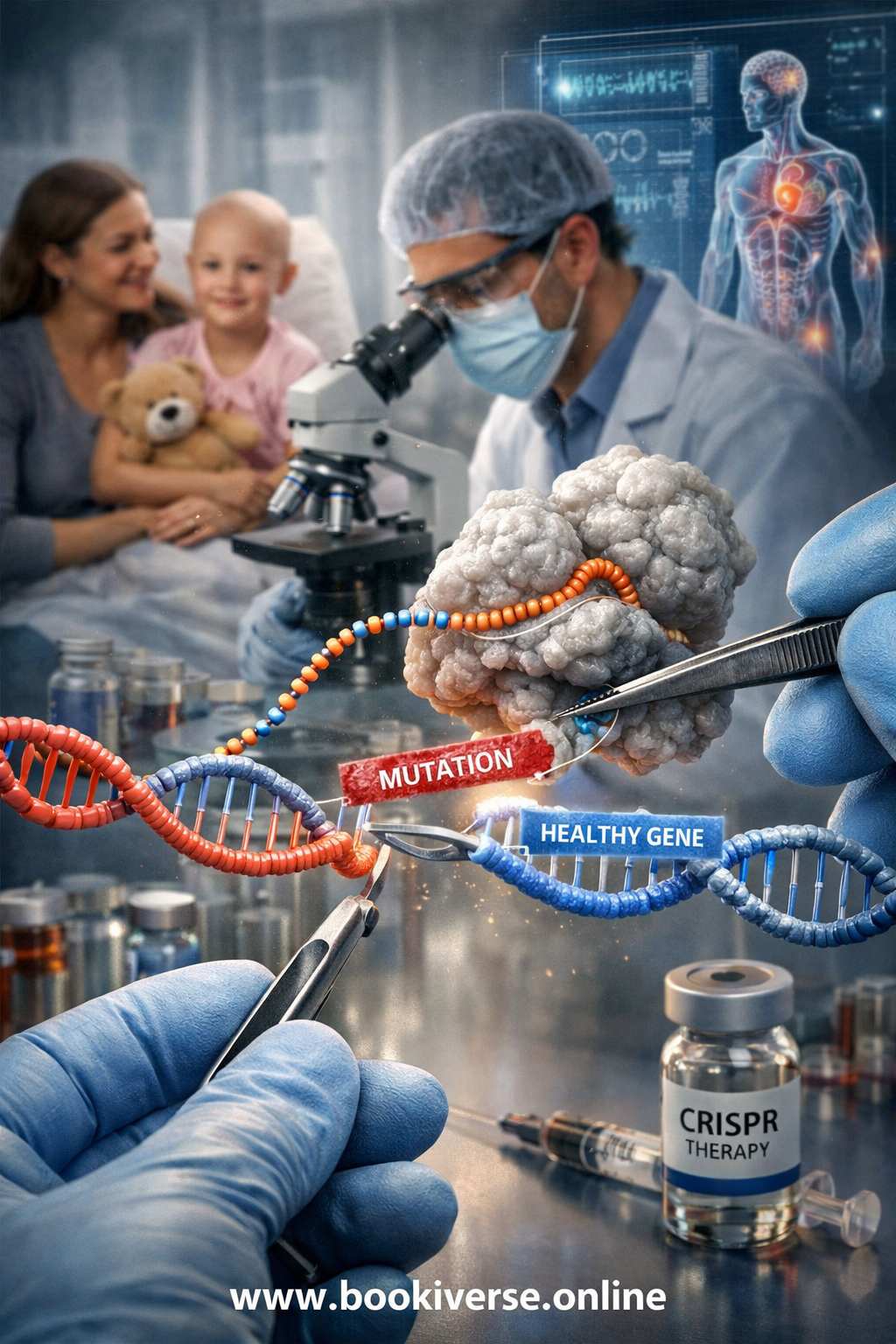
The elegance of the CRISPR-Cas9 system lies in its simplicity and programmability. It consists of two key components. The first is the Cas9 enzyme, which acts as a pair of "molecular scissors" capable of cutting DNA. The second is a piece of guide RNA (gRNA), which can be engineered in the laboratory to match a specific target sequence of DNA within a genome. When introduced into a cell, the gRNA guides the Cas9 enzyme to the precise location on the DNA that scientists wish to modify. Once there, Cas9 makes a clean cut across the double helix. The cell's natural DNA repair mechanisms then take over. Scientists can exploit this repair process in two ways: they can allow the cell to repair the break on its own, which often results in the deactivation of the targeted gene, or they can introduce a new, desired sequence of DNA that the cell can use as a template to patch the gap, effectively rewriting the genetic code. This versatility makes CRISPR a far more efficient and accessible tool than previous gene-editing techniques.
The Therapeutic Frontier and the Specter of Enhancement
The most celebrated promise of CRISPR is its potential to treat a vast range of genetic disorders. Somatic gene therapy—editing the genes of a patient's non-reproductive cells—is already showing remarkable progress in clinical trials for conditions like sickle cell anemia and certain types of blindness. By correcting the faulty gene in the affected cells, it is possible to alleviate or even cure the disease in that individual. This application is widely viewed as a laudable extension of medicine. The ethical landscape becomes substantially more complex, however, with the prospect of germline editing—modifying the DNA of embryos or reproductive cells. Such changes would be heritable, passed down through all subsequent generations. While this could theoretically eradicate hereditary diseases from a family's lineage forever, it also opens the door to non-therapeutic "enhancement." This raises the specter of a future where genetic modifications are used not just to prevent disease, but to select for desirable traits like intelligence, physical prowess, or appearance, potentially leading to unprecedented forms of social inequality and a redefinition of what is considered "natural."
Ecological and Unforeseen Consequences
The implications of CRISPR extend beyond human health into the broader ecosystem. The technology could be used to engineer crops for greater yield and resistance to drought and pests, which could be instrumental in addressing global food security. It also offers the possibility of "gene drives," a mechanism that can rapidly spread a specific genetic modification through an entire population of wild organisms. This could be used, for example, to eliminate malaria by altering mosquitoes so they can no longer carry the parasite. However, the power to permanently alter a species in the wild carries colossal risks. An engineered gene drive, once released, would be virtually impossible to recall. Any unforeseen negative consequences for the target species or the wider ecosystem—such as the collapse of a food web—could be irreversible. This necessitates an exceptionally high degree of caution and robust international governance to oversee any potential environmental applications. The power to edit the genome is not just the power to cure, but the power to fundamentally and permanently reshape the natural world, a responsibility of staggering proportions.